Hereditary hemochromatosis as an independent predictor of portal vein thrombosis: national inpatient sample (2016-2020) analysis
Hemocromatosis hereditaria como predictor independiente de trombosis de la vena porta: análisis de la muestra nacional de pacientes hospitalizados (2016-2020)
Kharel H1; Bhandari SK2; Pokhrel NB3; Kharel Z4; Jain T1; Sanjeevi A1; Niu C1
1 Department of Internal Medicine, Rochester General Hospital, New York, USA
2 Department of Internal Medicine, Tribhuvan University Teaching Hospital, Kathmandu, Nepal
3 Department of Internal Medicine, Norwalk Hospital, Connecticut, USA
4 Department of Hematology and Oncology, Rochester General Hospital, New York, USA
Himal Kharel1 (https://orcid.org/0000-0002-6794-8352)
Samikchhya Keshary Bhandari2 (https://orcid.org/0000-0001-6817-518X)
Nishan Babu Pokhrel3 (https://orcid.org/0000-0002-4278-5753)
Zeni Kharel4 (https://orcid.org/0000-0002-5564-3746)
Tripti Jain1 (https://orcid.org/0009-0000-9240-2055)
Aditya Sanjeevi1 (https://orcid.org/0000-0001-8899-633X)
Chengu Niu1 (https://orcid.org/0000-0001-5610-5897)
Keywords: hereditary hemochromatosis,
portal vein thrombosis,
iron overload,
national inpatient sample.
Palabras claves: hemocromatosis hereditaria,
trombosis de la vena porta,
sobrecarga de hierro,
muestra nacional de pacientes hospitalizados.
Abstract
Introduction. Hereditary hemochromatosis is a common inherited metabolic disorder of iron metabolism with variable penetrance. Portal vein thrombosis has several well-known causes including cirrhosis, myeloproliferative neoplasm, inflammatory conditions of the abdomen, intra-abdominal malignancy, intra-abdominal surgery, and thrombophilia. It is unknown whether hereditary hemochromatosis is associated with portal vein thrombosis.
Material and methodology. A retrospective analysis of the National Inpatient Sample database from 2016 to 2020 was conducted using International Classification of Diseases-10 codes to identify hospitalizations with portal vein thrombosis. We used multivariate regression analysis to calculate the adjusted odds ratio after controlling for age, gender, insurance type, Charlson Comorbidity Index, cirrhosis, myeloproliferative neoplasm, inflammatory conditions of the abdomen, intra-abdominal malignancy, intra-abdominal surgery, history of past thrombosis, thrombophilia, end-stage renal disease, obesity, smoking, and hyperlipidemia.
Results. There were 25805 hospitalizations with primary diagnosis of portal vein thrombosis. Multivariate logistic regression showed that hereditary hemochromatosis was independently associated with portal vein thrombosis (Odds ratio= 4.7, 95% CI=1.15 to 19.35, p-value=0.03) when cirrhosis, myeloproliferative neoplasm, inflammatory conditions of the abdomen, intra-abdominal malignancy, history of thrombosis, history of abdominal surgery, history of thrombophilia, and end-stage renal disease among other factors were controlled.
Resumen
Introducción. La hemocromatosis hereditaria es un trastorno metabólico hereditario común del metabolismo del hierro con penetrancia variable. La trombosis de la vena porta tiene varias causas bien conocidas que incluyen cirrosis, neoplasia mieloproliferativa, afecciones inflamatorias del abdomen, neoplasia maligna intraabdominal, cirugía intraabdominal y trombofilia. Se desconoce si la hemocromatosis hereditaria está asociada con la trombosis de la vena porta.
Material y metodología. Se realizó un análisis retrospectivo de la base de datos de la Muestra Nacional de Pacientes Hospitalizados de 2016 a 2020 utilizando los códigos de la Clasificación Internacional de Enfermedades-10 para identificar hospitalizaciones con trombosis de la vena porta. Utilizamos un análisis de regresión multivariado para calcular el odds ratio ajustado después de controlar por edad, sexo, tipo de seguro, índice de comorbilidad de Charlson, cirrosis, neoplasia mieloproliferativa, afecciones inflamatorias del abdomen, neoplasia maligna intraabdominal, cirugía intraabdominal, antecedentes de trombosis pasada, trombofilia, enfermedad renal terminal, obesidad, tabaquismo e hiperlipidemia.
Resultados. Hubo 25.805 hospitalizaciones con diagnóstico primario de trombosis de la vena porta. La regresión logística multivariada mostró que la hemocromatosis hereditaria se asoció de forma independiente con la trombosis de la vena porta (odds ratio = 4.7, IC del 95 % = 1.15 a 19.35, valor de p = 0,03) cuando se controlaron cirrosis, neoplasia mieloproliferativa, afecciones inflamatorias del abdomen, neoplasia maligna intraabdominal, antecedentes de trombosis, antecedentes de cirugía abdominal, antecedentes de trombofilia y enfermedad renal terminal, entre otros factores.
Introduction
Hereditary hemochromatosis (HH) has a prevalence of 1 in 220 to 250 people(1). It is a common genetic disorder with variable penetrance. It is especially common in non-Hispanic whites of northern European origin. The clinical features of those affected include liver cirrhosis, diabetes mellitus, hypogonadism, cardiomyopathy, and arthropathy(1).
The lifetime risk of portal vein thrombosis in the general population is estimated to be around 1%(2). There are many well-known risk factors of portal vein thrombosis including, but not limited to, myeloproliferative neoplasm, inflammatory conditions of the abdomen (acute appendicitis, acute diverticulitis, and acute pancreatitis), intra-abdominal malignancy and intra-abdominal surgery(3).
Gill et al found that one standard deviation increase in ferritin was associated with a significantly increased risk of venous thromboembolism with an odds ratio(OR) of 1.92(4). It is unknown if hereditary hemochromatosis is independently associated with the development of portal vein thrombosis.
Methods
Data source
We used the National Inpatient Sample (NIS) database from 2016 to 2020, as the source of data(5). This database, part of the Healthcare Cost and Utilization Project (HCUP) and maintained by the Agency for Healthcare Research and Quality (AHRQ), represents a weighted 20% sample of all hospital discharges. It includes demographic, economic, and clinical data.
Ethical considerations
Due to the de-identified nature of the patient information contained within, obtaining institutional review board approval was not necessary.
Variables and outcomes
We utilized International Classification of Disease-10 (ICD-10) codes to identify portal vein thrombosis (I81) as the primary diagnosis. Secondary diagnoses included a variety of conditions, each identified by specific ICD-10 codes: cirrhosis (K7030, K7031, K7460, K745, K7469), myeloproliferative neoplasms (D471, D473), and inflammatory abdominal conditions such as acute appendicitis, diverticulitis, and pancreatitis (K35200 to K35219, K3530 to K3533, K3580, K35890, K35891, K8500 to K8512, K8520, K5720 to K5733), intra-abdominal malignancies (C180 to C20, C220, C250 to C259), intra-abdominal surgeries (0D, 0F), history of thrombosis (Z86718), thrombophilia (D685, D686), end-stage renal disease (N186), obesity (E66), smoking (F1721), and hyperlipidemia (E7800, E7801, E782 to E785). The primary outcome investigated was the association between hereditary hemochromatosis and portal vein thrombosis, expressed as an odds ratio.
Statistical analysis
Statistical analysis was performed with STATA version SE18.0. We executed a multivariate regression analysis, adjusting for factors like age, gender, primary payer, Charlson Comorbidity Index, cirrhosis, myeloproliferative neoplasm, and inflammatory abdominal conditions (such as acute appendicitis, diverticulitis, and pancreatitis), as well as intra-abdominal malignancy, intra-abdominal surgery, previous thrombosis, thrombophilia, end-stage renal disease, obesity, smoking, and hyperlipidemia, to compute the adjusted odds ratio (OR).
Results
Baseline characteristics
During the 5-year period from 2016 to 2020, there were 25805 hospitalizations with a primary diagnosis of portal vein thrombosis. The mean age was 54.6 years (95% confidence interval 54.1 to 55.1). Out of them, 45.4% were female (95% confidence interval 44.1 to 46.8). The majority of patients were white (69.6%) followed by Asian (12.2%), and black (12%). Most patients were on private insurance (40.4%) and Medicare (35.5%). The majority (92%) of patients had a Charlson co-morbidity index of greater than or equal to 3.
Multivariate analysis
Multivariate logistic regression analysis revealed that hereditary hemochromatosis independently correlates with an increased risk of portal vein thrombosis, with an odds ratio (OR) of 4.7 (95% confidence interval (CI) ranging from 1.15 to 19.35, p-value of 0.03). This finding was established after adjusting for other risk factors such as cirrhosis, myeloproliferative neoplasm, inflammatory abdominal conditions, intra-abdominal malignancy, previous thrombosis, past abdominal surgery, thrombophilia, and end-stage renal disease, among others. The findings are summarized in table 1 and figure 1.
|
|
Odds ratio of portal vein thrombosis |
95% confidence interval |
P-value |
|
Risk factors |
|||
|
Hereditary hemochromatosis |
4.72 |
1.15- 19.35 |
0.03 |
|
Cirrhosis |
11.2 |
9.4- 13.3 |
0.00 |
|
Myeloproliferative neoplasm |
5.5 |
3.55 -8.56 |
0.00 |
|
Inflammatory conditions of abdomen |
2.5 |
1.8 -3.4 |
0.00 |
|
History of abdominal malignancy |
8.9 |
7.1- 11.3 |
0.00 |
|
History of prior thrombosis |
2.54 |
2 - 3.22 |
0.00 |
|
History of abdominal surgery |
1.8 |
1.5 - 2.13 |
0.00 |
|
History of thrombophilia |
8.8 |
6.9 – 11.2 |
0.00 |
|
End stage renal disease |
0.14 |
0.05 - 0.35 |
0.00 |
|
Obesity |
1.28 |
1.07 - 1.5 |
0.02 |
|
Smoking |
1.1 |
0.9 - 1.3 |
0.29 |
|
Hyperlipidemia |
0.72 |
0.6 - 0.86 |
0.00 |
|
Demographics |
|||
|
Female |
0.78 |
0.68 - 0.9 |
0.00 |
|
Age |
1 |
0.99-1 |
0.9 |
|
Charlson Co-morbidity Index |
|||
|
1 |
1.21 |
0.94 - 1.5 |
0.13 |
|
2 |
0.97 |
0.72 - 1.31 |
0.88 |
|
>3 |
1.38 |
1.08 - 1.7 |
0.01 |
|
Median household income |
|||
|
2nd quartile |
1.03 |
0.85 - 1.25 |
0.14 |
|
3rd quartile |
1.17 |
0.96 – 1.42 |
0.00 |
|
4th quartile |
1.09 |
0.89 – 1.34 |
0.00 |
|
Race |
|||
|
Black |
0.96 |
0.78- 1.2 |
0.77 |
|
Hispanic |
1.08 |
0.9 -1.34 |
0.44 |
|
Asian |
0.9 |
0.58 - 1.4 |
0.6 |
|
Native American |
0.69 |
0.28- 1.67 |
0.3 |
|
Other |
1.40 |
1 – 1.96 |
0.05 |
Table 1. Multivariate logistic regression findings
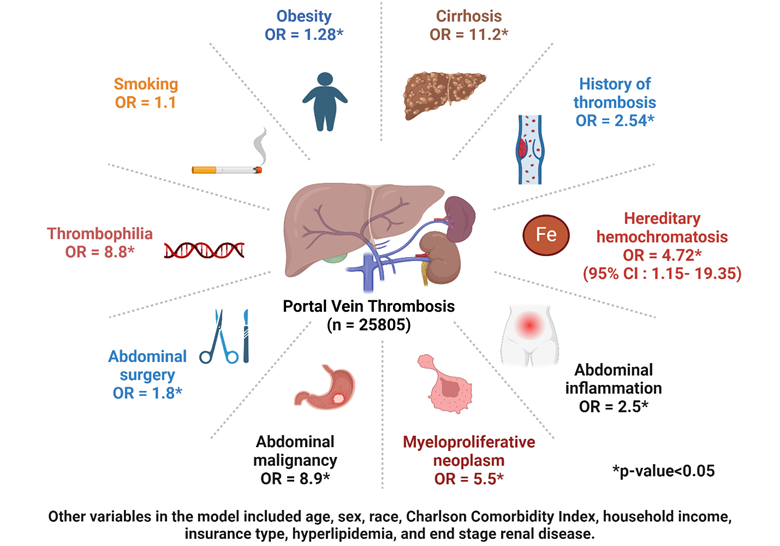
Figure 1. Findings of multivariate analysis
Discussion
Our study revealed that hereditary hemochromatosis is independently linked to an elevated risk of portal vein thrombosis, even when accounting for other recognized risk factors such as cirrhosis, myeloproliferative neoplasm, inflammatory abdominal conditions, intra-abdominal malignancy, previous thrombosis, past abdominal surgery and thrombophilia. This finding is noteworthy, as hereditary hemochromatosis has not been previously identified as a cause of portal vein thrombosis.
In a Swedish autopsy study of 24 000 autopsies, about 14% were found to have idiopathic portal vein thrombosis, and the rest of them were found to be caused by cirrhosis, primary or secondary hepatobiliary malignancy, major infectious or inflammatory abdominal disease, or myeloproliferative disease(2). Notably, hereditary hemochromatosis, particularly prevalent among non-Hispanic whites of northern European descent, was not considered in this study.
A large-scale Mendelian Randomization study in 2019 found that iron overload significantly increases risk of venous thromboembolism with OR of 1.37(4) which was consistent with another case control study which showed dose-dependent relationship between hepcidin levels and risk of venous thromboembolism(6).
Iron overload has been shown to enhance thrombus formation post-vascular injury, elevate vascular reactive oxygen species (ROS) production, and impair endothelial-dependent vasorelaxation in mice(7). In the same study, administration of ROS scavenger reversed the effect. Iron induced oxidative stress decreases bioavailability of nitric oxide, leading to endothelial dysfunction and platelet activation(8,9). Given the susceptibility of fibrinogen to oxidation, exposure of fibrinogen to ferric ion promotes fibrin formation, promotes platelet aggregation and causes less efficient plasminogen activation by tissue-type plasminogen activator(10,11). All these factors would explain the increased risk of thrombosis in setting of iron overload. These findings are summarized in figure 2.
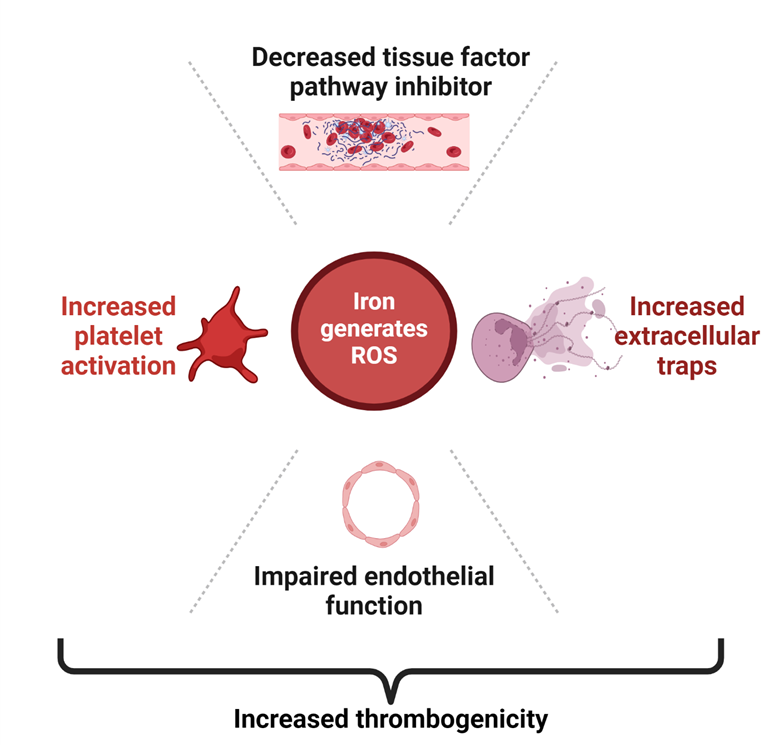
Figure 2. Potential pathophysiological mechanism of hypercoagulability in hereditary hemochromatosis
We were able to find only a single case report from France where compound heterozygous C282Y/H63D hereditary hemochromatosis was implicated in a 49-year-old male presenting with idiopathic portal vein thrombosis and without cirrhosis(12).
Hereditary hemochromatosis may have association with factor V Leiden mutation. An observation study of a sample of unscreened patient with thrombosis by Xie et al. in 1998(13) showed that frequency of C282Y heterozygotes was significantly higher in factor V Leiden carriers than in factor V Leiden-negative patients. It is possible that a minor dysregulation of iron metabolism in the setting of presence of heterozygous state for factor V Leiden could have increased risk of thrombosis. However, our study accounted for thrombophilias in the multivariate analysis.
Our study's limitations include its retrospective nature, which introduces potential bias, and the principle that correlation does not equate to causation. Additionally, as the national inpatient sample is an administrative database, it is prone to coding inaccuracies and data mischaracterization. Specific ICD-10 codes for splenic and mesenteric vein thrombosis were absent, excluding them from our analysis. Future prospective studies are warranted to confirm this association.
Author contribution
HK came up with the idea. HK and SKB did the statistical analysis. ZK made the figure. NBP, TJ, AS, and CN wrote the manuscript. The authors have made a substantial contribution to the conception or design of the work, and data acquisition, analysis, or interpretation. They have participated in the article drafting or the critical revision of its intellectual content. They have agreed to the final version of the manuscript and can defend every aspect of the manuscript to guarantee that all the questions related to the accuracy or integrity of its content have been appropriately investigated and resolved.
References
1. Bacon BR, Adams PC, Kowdley KV, Powell LW, Tavill AS. Diagnosis and management of hemochromatosis: 2011 Practice Guideline by the American Association for the Study of Liver Diseases. Hepatology. 2011;54(1):328-343. doi:10.1002/hep.24330
2. Ögren M, Bergqvist D, Björck M, Acosta S, Eriksson H, Sternby NH. Portal vein thrombosis: Prevalence, patient characteristics and lifetime risk: A population study based on 23 796 consecutive autopsies. World J Gastroenterol. 2006;12(13):2115-2119. doi:10.3748/wjg.v12.i13.2115
3. Quarrie R, Stawicki SP. Portal vein thrombosis: What surgeons need to know. Int J Crit Illn Inj Sci. 2018;8(2):73-77. doi:10.4103/IJCIIS.IJCIIS_71_17
4. Gill D, Brewer CF, Monori G, et al. Effects of Genetically Determined Iron Status on Risk of Venous Thromboembolism and Carotid Atherosclerotic Disease: A Mendelian Randomization Study. Journal of the American Heart Association. 2019;8(15):e012994. doi:10.1161/JAHA.119.012994
5. HCUP-US NIS Overview. Accessed December 19, 2023. https://hcup-us.ahrq.gov/nisoverview.jsp
6. Ellingsen TS, Lappegård J, Ueland T, Aukrust P, Brækkan SK, Hansen JB. Plasma hepcidin is associated with future risk of venous thromboembolism. Blood Adv. 2018;2(11):1191-1197. doi:10.1182/bloodadvances.2018018465
7. Day SM, Duquaine D, Mundada LV, et al. Chronic iron administration increases vascular oxidative stress and accelerates arterial thrombosis. Circulation. 2003;107(20):2601-2606. doi:10.1161/01.CIR.0000066910.02844.D0
8. Cooke JP. Does ADMA Cause Endothelial Dysfunction? Arteriosclerosis, Thrombosis, and Vascular Biology. 2000;20(9):2032-2037. doi:10.1161/01.ATV.20.9.2032
9. Kenet G, Freedman J, Shenkman B, et al. Plasma glutathione peroxidase deficiency and platelet insensitivity to nitric oxide in children with familial stroke. Arterioscler Thromb Vasc Biol. 1999;19(8):2017-2023. doi:10.1161/01.atv.19.8.2017
10. Feng YH, Hart G. In vitro oxidative damage to tissue-type plasminogen activator: a selective modification of the biological functions. Cardiovasc Res. 1995;30(2):255-261.
11. Upchurch GR, Ramdev N, Walsh MT, Loscalzo J. Prothrombotic Consequences of the Oxidation of Fibrinogen and their Inhibition by Aspirin. J Thromb Thrombolysis. 1998;5(1):9-14. doi:10.1023/A:1008859729045
12. Delval L, Brohée D, Cauchie P, Vanhaeverbeek M, Hilbert P. [Acute portal thrombosis revealing hereditary hemochromatosis: report of a case]. Rev Med Brux. 2002;23(2):83-86.
13. Xie YG, Lillicrap DP, Taylor SA. An association between the common hereditary hemochromatosis mutation and the factor V Leiden allele in a population with thrombosis. Blood. 1998;92(4):1461-1462.