Near missed aggressive natural killer cell leukaemia – importance of extensive immunophenotyping
Leucemia agresiva de células natural killer: importancia de la inmunofenotipificación
Kunnumbrath A1, Azad S1, Arya S1, Agarwal M2, Balasubramanian P1
1 Department of Pathology, All India Institute of Medical Sciences (AIIMS), Rishikesh, India
2 Department of General medicine, All India Institute of Medical Sciences, Rishikesh, India, PIN-249203
ORCID:
Dr Arathi kunnumbrath: https://orcid.org/0000-0001-8718-1622
Dr Shweta Azad: https://orcid.org/0000-0001-9280-5831
Dr Shubhangee Arya: https://orcid.org/0009-0006-7324-9212
Dr Mayank Agarwal: https://orcid.org/0000-0001-7895-3440
Dr Priyavadhana Balasubramanian: https://orcid.org/0000-0002-4137-8056
Palabras claves: leucemia agresiva de células natural killer,
citometría de flujo,
inmunofenotipificación.
Keywords: aggressive natural killer leukaemia,
flow cytometry,
immunophenotyping.
Abstract
Aggressive natural killer cell leukaemia (ANKL) is a rare neoplasm of NK cells with poor prognosis, varied morphology and positivity for CD2, CD3 epsilon and CD56, negative for surface CD3 and CD5. A 32-year-old man presented with high grade fever, generalized weakness, significant weight loss in the last 2 months and a lower limb mass, diagnosed elsewhere as Ewing sarcoma. He initially presented with TLC of 8000/uL which drastically increased to 52000/uL, with presence of atypical lymphoid cells. Bone marrow aspiration and biopsy was performed, bone marrow aspirate showed 44% atypical lymphoid cells that were 1.5 to 2 times the size of red blood cells with scant blue agranular cytoplasm, clumped chromatin and inconspicuous nucleoli, and flow cytometric immunophenotyping showed atypical lymphoid cells that were positive for CD45, CD38 and CD 56 and negative for CD34, CD3, CD4, CD5, CD7, CD8, CD20, CD10, CD19, CD138, kappa and lambda. Since a definitive diagnosis could not be reached on this, IHC on bone marrow biopsy was done, which showed the atypical lymphoid cells to be positive for CD45, CD3, CD2, CD56, CD38 and negative for CD117, CD5, CD7, CD30, CD20, CD57 and EBV. A diagnosis of aggressive natural killer lymphoma was reached. The diagnosis was difficult as the morphology and flow cytometry did not show any specific features of ANKL. The diagnosis was made after extensive immunophenotyping on bone marrow biopsy. This highlights the importance of doing NK cell markers when basic panel of antibodies are not supportive.
Resumen
La leucemia agresiva de células natural killer (ANKL) es una neoplasia poco frecuente de células NK con mal pronóstico, morfología variada y positividad para los CD2, CD3 épsilon y CD56, negativa para los CD3 y CD5 de superficie. Un hombre de 32 años presentó fiebre alta, debilidad generalizada, pérdida de peso significativa desde hace 2 meses y una masa en las extremidades inferiores, diagnosticada en otro lugar como sarcoma de Ewing. Inicialmente presentó leucocitos de 8000/uL, que aumentó drásticamente a 52000/uL, con presencia de células linfoides atípicas. Se realizó una aspiración y una biopsia de médula ósea. El aspirado de médula ósea mostró un 44% de células linfoides atípicas que tenían entre 1,5 y 2 veces el tamaño de los glóbulos rojos con escaso citoplasma agranular azul, cromatina agrupada y nucléolos discretos, y la inmunofenotipificación por citometría de flujo mostró células linfoides atípicas que eran positivas para CD45, CD38 y CD 56 y negativas para CD34, CD3 y CD4. CD5, CD7, CD8, CD20, CD10, CD19, CD138, kappa y lambda.
Como no se pudo llegar a un diagnóstico definitivo al respecto, se realizó un estudio inmunohistoquímico de la biopsia de médula ósea, que mostró que las células linfoides atípicas eran positivas para CD45, CD3, CD2, CD56, CD38 y negativas para CD117, CD5, CD7, CD30, CD20, CD57 y EBV. Se llegó a un diagnóstico de linfoma agresivo de células natural killer. El diagnóstico fue difícil ya, que la morfología y la citometría de flujo no mostraron ninguna característica específica de la ANKL. El diagnóstico se realizó después de una inmunofenotipificación exhaustiva mediante una biopsia de médula ósea. Esto pone de relieve la importancia de utilizar marcadores de células NK cuando el panel básico de anticuerpos no es suficiente.
Case report
Introduction
Aggressive natural killer cell leukaemia (ANKL) is a rare neoplasm of NK cell origin with very poor prognosis, with less than 3 months median survival after diagnosis(1,2). It accounts to 10% of all large granular lymphocyte (LGL) disorders and is typically seen in young adult males. Patients present with fever, fatigue, jaundice, lymphadenopathy and organomegaly, with some cases having disseminated intravascular coagulation and related symptoms(1,2). ANKL cells show a varied morphology, from large granular lymphocyte like appearance to large atypical pleomorphic cells(1,2). The usual immunoexpression shows positivity for CD2, CD3 epsilon and CD56, negative for surface CD3 and CD5, with noted aberrancies like loss of expression of CD2, CD7 or CD45(1). The diagnosis of ANKL can be challenging due to the varied morphology. Here, we report a case of ANKL with diagnostic dilemma on clinical presentation and morphological features.
Case presentation
A 32-year-old man presented with symptoms of high-grade fever, generalized weakness and significant weight loss in the last 2 months. On physical examination, he had mild pallor, bilateral pedal edema, hepatosplenomegaly and palpable right cervical and left inguinal lymph nodes. He also had a mass in left leg for which surgical excision was done in another hospital. His initial complete blood counts (CBC) showed bicytopenia (haemoglobin of 7.1 g/dL, total leucocyte counts (TLC): 8,000/cu.mm, platelet count: 17,000/cu.mm. His TLC increased drastically to 52,000/cu.mm in two days with 68% atypical lymphoid cells. The following biochemical parameters were elevated: lactate dehydrogenase 1450 U/L (0 -248 U/L), high-sensitivity C-reactive protein 25.5 mg/L (0-1 mg/L), procalcitonin 14.0 ng/mL (0 - 0.5 ng/mL), ferritin > 1650.0 ng/mL (22-322 ng/mL), fibrinogen > 1200 mg/dL (200-400 mg/dL) and prothrombin time > 120 (12-14 seconds). Liver function tests and renal function tests were mildly deranged. Ultrasonography of bilateral limb showed no evidence of thrombosis. Magnetic resonance imaging of the leg mass done outside showed a hyperintense cystic lesion measuring 4.2x2.7x2.2 cm in subcutaneous plane in posterolateral aspect of distal leg-likely to be benign in nature and the outside histopathology diagnosis was suggestive of small round cell tumour, possibly Ewing sarcoma. The patient had features of sepsis with acute liver and kidney injury, coagulopathy, bicytopenia and pulmonary edema.
For further evaluation, bone marrow aspiration and biopsy were done, which showed 44% atypical lymphoid cells that were 1.5 to 2 times the size of red blood cells with scant blue agranular cytoplasm, nuclei with clumped chromatin and inconspicuous nucleoli. Occasional macrophages were showing hemophagocytosis. Acute leukaemia or Ewing sarcoma metastasis from bone was considered as the possibilities. Flow cytometry immunophenotyping (FCI) showed atypical lymphoid cells that were positive for CD45, CD38 and CD 56 and negative for CD34, CD3, CD4, CD5, CD7, CD8, CD20, CD10, CD19, CD138, kappa and lambda (Figure 1). Based on these markers, the diagnosis of Ewings sarcoma metastasis was ruled out and the diagnosis of a lymphoproliferative neoplasm was confirmed, further categorization of which was done on bone marrow biopsy. Bone marrow biopsy showed scattered interstitial and small clusters of atypical medium sized lymphoid cells with scant to moderate amount of cytoplasm, fine chromatin and one to two prominent nucleoli. On immunohistochemistry (IHC), these cells were positive for CD45, CD3, CD2, CD56, CD38 and negative for CD117, CD5, CD7, CD30, CD20, CD57 and EBV (Figure 2). Initially, the possibility of NK cell lineage neoplasm was considered only based on CD56 positivity in FCI as the morphology was not suggestive of NK cell origin tumour. The final diagnosis of aggressive natural killer cell leukaemia was made based on bone marrow morphology, flow cytometric immunophenotyping and immunohistochemistry.
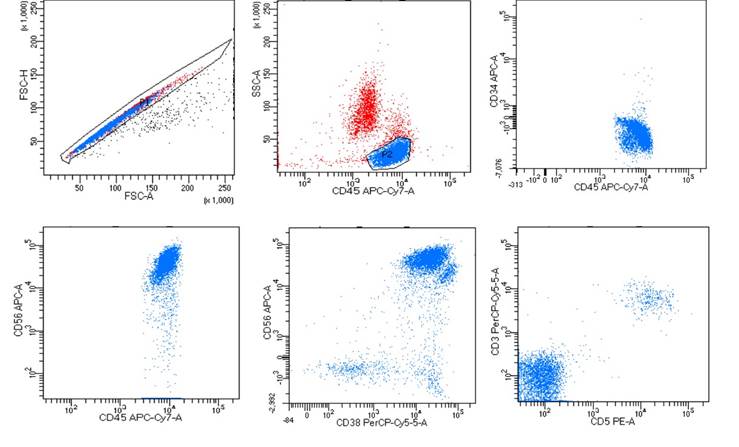
Figure 1. Flow cytometric immunophenotyping of the bone marrow aspirate identified 75% atypical lymphoid cells. A) FSC-H FSC-A plot to rule out doublets, B) cells of interest with bright CD45 gated as P2. Cells of interest are C) negative for CD34, D) positive for CD56, E) positive for CD38 and F) negative for CD5 and CD3.
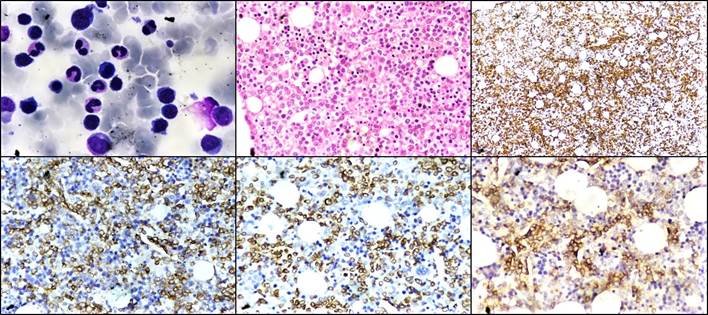
Figure 2. (A) Bone marrow aspirate showing atypical lymphoid cells (Giemsa stain, ×400). (B) Bone marrow biopsy showing diffuse infiltration by these infiltrative atypical lymphoid cells (H&E stain, ×400) (C) Immunohistochemistry CD45 (IHC, ×100). (D) Immunohistochemistry CD2 (IHC, ×400). (E) Immunohistochemistry CD3 (IHC, ×400). (F) Immunohistochemistry CD56 (IHC, ×400)
Patient was in sepsis and treatment was started with multiple antibiotics (piperacillin/tazobactam, clindamycin, meropenem), intravenous fluids and non-adrenaline. The patient died of septic shock on 11th day of admission in spite of getting continuous oxygen support.
Discussion
ANKL is a NK cell neoplasm that is more prevalent among Asians, with an aggressive clinical course and an association with EBV in around 80% cases. The patients are usually young adult males, like our patient. EBV might have a role in the pathogenesis, however it is not well understood(1-3). In our case, EBV against EBV-LMP antigen done by immunohistochemistry was negative. The atypical cells morphologically resemble large granular lymphocytes with basophilic cytoplasm having azurophilic granules[4]; however, cases showing large atypical pleomorphic tumour cells are also reported(2). In our case, the classical morphology with large granular lymphocyte like appearance was not seen.
NK cells are positive for CD2, CD3 epsilon, CD16 and CD56, negative for surface CD3, myeloid and B-cell markers. Unlike T-cell large granular lymphocytes, NK cells are negative for CD3(1,4,5). The clonal rearrangement of T-cell receptor (TCR) genes is also not seen in NK cells. The atypical cells in ANKL can be CD3 positive by immunohistochemistry on paraffin-embedded tissue, as it contains CD3 epsilon specific polyclonal antibodies and natural killer T cells have CD3 epsilon subunits in their cytoplasm(6).
ANKL was diagnosed in our case based on the immunoexpression assessed using flow cytometry and immunohistochemistry. In hindsight, the clinical manifestations were also supportive, which included fever, cytopenia, hepatosplenomegaly, and the biochemical parameters were suggestive of disseminated intravascular coagulation. In addition to this, the patient had a thigh mass, which is not usually seen in the clinical presentation of ANKL, which led us to include bone marrow metastasis in the differential diagnosis. Morphologically, the atypical cells looked lymphoid in nature, so a lymphoid neoplasm was also considered; however, due to the drastic increase in TLC and poor clinical response of the patient, a diagnosis of acute leukaemia was favored before the flow cytometry was done. On flow cytometry, these atypical cells were found to be lymphoid and lacked expression of immaturity markers. T-cell LGL leukaemic cells typically co-express CD3+, CD8+ and CD57+ markers(1), so the CD8 and CD57 negativity excluded T-cell large granular lymphocytic leukaemia in our case. Blastic plasmacytoid dendritic cell leukaemia was excluded with CD4 negativity. CD3 can also be positive in NK cell neoplasms though cytoplasmic CD3 is lineage specific for T cells(6). The clear expression of CD56 and CD2 in our case helped us in making a diagnosis of ANKL more likely.
The literature mentions quite varied morphology and clinical course in ANKL, in addition to the specific symptoms and morphology(2,6). Reports of delayed diagnosis of ANKL due to non-specific clinical symptoms and fewer tumour cells in the peripheral blood and/or bone marrow on presentation, as seen in our case, have already been reported in literature(7).
ANKL is a potentially chemo sensitive disease, and complete remission might be achieved on treatment with combined immunotherapy including L-asparginase. Allogeneic hematopoietic cell transplantation can also achieve a complete response and potential cure for some patients(2,7). However, the prognosis of ANKL is still quite poor, with a median survival shorter than one year(5).
A range of cytogenetic abnormalities has been reported in cases of ANKL, such as del(6)(q21q25), del(11q), i(7)(q10), losses in 7p and 17p, and gains in 1q. TP53 is found to be mutated in one third of the cases, with mutations in genes of JAK/STAT and RAS/MAPK pathways and immune checkpoint molecules also being observed in some cases(1).
Informed consent has been obtained from the patient’s family for this publication, and no images where patient’s identity will be disclosed is being used. No funding has been received.
Conclusion
We have reported a case of ANKL with non-specific presentation. This case becomes interesting, as the initial morphology was not suggestive of NK cells and showed medium sized cells with scant, agranular cytoplasm and inconspicuous nucleoli. The CD56 expression, revealed in flow cytometry, led us to consider the possibility of a NK cell origin tumour. This highlights the importance of doing NK cell markers when the basic panel of antibodies are not supportive.
Authors' contribution: the authors have made a substantial contribution to the conception or design of the work, and data acquisition, analysis, or interpretation. They have participated in the article drafting or the critical revision of its intellectual content. They have agreed to the final version of the manuscript and can defend every aspect of the manuscript to guarantee that all the questions related to the accuracy or integrity of its content have been appropriately investigated and resolved.
References
1. Swerdlow SH, Campo E, Harris NL, Jaffe ES, Pileri SA, Stein H et al: WHO Classification of Tumours of Haematopoietic and Lymphoid Tissues (Revised 4th edition). IARC: Lyon 2017.
2. Ishida F. Recent progress of diagnosis and treatment in NK cell neoplasms. Rinsho Ketsueki. 2015 Jun;56(6):645-50. DOI: 10.11406/rinketsu.56.645
3. Mori N, Yamashita Y, Tsuzuki T, Nakayama A, Nakazawa M, Hasegawa Y et al. Lymphomatous features of aggressive NK cell leukaemia/lymphoma with massive necrosis, haemophagocytosis and EB virus infection. Histopathology. 2000 Oct;37(4):363-71. DOI: 10.1046/j.1365-2559.2000.00936.x
4. Hu Y, Wang J. Aggressive natural killer cell leukemia: a case report. Int Med Case Rep J.2017; 10:389-91. DOI: 10.2147/IMCRJ.S136148
5. Ishida F, Ko YH, Kim WS, Suzumiya J, Isobe Y, Oshimi K et al. Aggressive natural killer cell leukemia: therapeutic potential of Lasparaginase and allogeneic hematopoietic stem cell transplantation. Cancer Sci. 2012 Jun;103(6):1079-83. DOI: 10.1111/j.1349-7006.2012.02251.x
6. Ruskova A, Thula R, Chan G. Aggressive Natural Killer-Cell Leukemia: report of five cases and review of the literature. Leuk Lymphoma. 2004 Dec;45(12):2427-38. DOI: 10.1080/10428190400004513
7. Boysen AK, Jensen A, Johansen P, Dybkær K, Nyegaard M. Treatment of Aggressive NK-Cell Leukemia: A Case Report and Review of the Literature. Case reports in hematology. 2011. DOI: 10.1155/2011/818469
Figure 1. Flow cytometric immunophenotyping of the bone marrow aspirate identified 75% atypical lymphoid cells. A) FSC-H FSC-A plot to rule out doublets, B) cells of interest with bright CD45 gated as P2. Cells of interest are C) negative for CD34, D) positive for CD56, E) positive for CD38 and F) negative for CD5 and CD3.
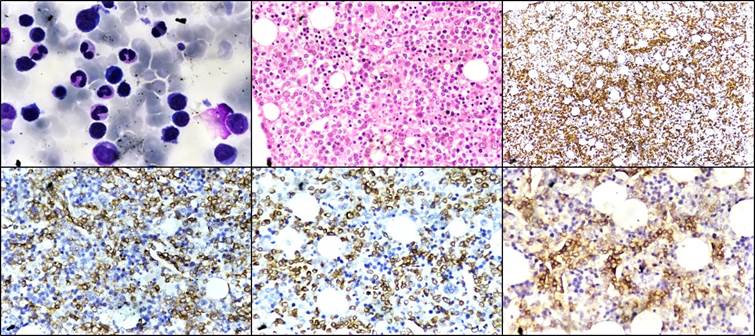
Figure 2. (A) Bone marrow aspirate showing atypical lymphoid cells (Giemsa stain, ×400). (B) Bone marrow biopsy showing diffuse infiltration by these infiltrative atypical lymphoid cells (H&E stain, ×400) (C) Immunohistochemistry CD45 (IHC, ×100). (D) Immunohistochemistry CD2 (IHC, ×400). (E) Immunohistochemistry CD3 (IHC, ×400). (F) Immunohistochemistry CD56 (IHC, ×400).